The Energy of Activation Is Best Described as
Decreasing the activation energy. 1energy input needed to break bonds of reactants.

Activation Energy Definition Formula Si Units Examples Calculation
Which best describes the energy of activation.
. A rapid reaction happens very rapidly as it has very low activation energy. A protein that speeds up a chemical reaction. B the minimum kinetic energy that particles must possess for a chemical reaction to occur.
None of the answer choices are correct. KJ and the reaction is D. KJ and the reaction is C.
The best definition of activation energy is that activation energy is the energy required to bind a substrate to an active site option C. 2energy stored in chemical bonds. A The greater the activation energy the greater the enthalpy of reaction.
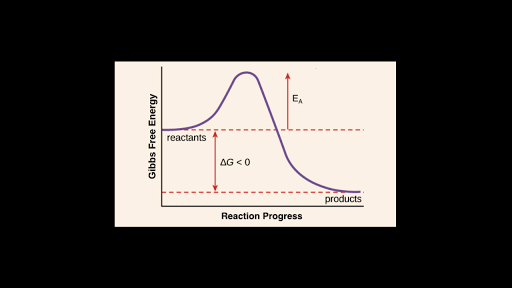
The activation energy for a reaction is illustrated in the potential energy diagram by the height of the hill between the reactants and the products. It is lower if the reaction temperature decreases. If the activation energy is low rate will be high.
And if the energy of the colliding molecules is less than the activation energy no reaction will also occur. 5Higher activation energy results in a slower reaction rate. What is the relationship between activation energy and rate constant.
The activation energy is 50. The activation energy is 10. The activation energy is 10.
The difference in energy between reactants and products. The energy level of the products. A numerical description of the amount of energy needed by colliding reactant molecules in order to form.
D the energy required to separate ions in a crystalline solid. KJ and the reaction is endothermic. What is enzyme activation energy.
The higher the activation energy the slower the chemical reaction will be. A type of exergonic reaction b. Which of the following best describes the activation energy of a chemical reaction.
B The amount of energy gained in an endothermic reaction c All of the above d The amount of energy required to start a reaction. Why is activation energy important. Up to 24 cash back Which statement correctly describes the energy changes that occur in the forward reaction.
1 on a question. The energy level of the reactants. The activation energy in the Arrhenius equation can best be described as asked Jun 26 2017 in Chemistry by maju88 a.
As well it mathematically expresses the relationships we established earlier. If there is no activation energy no reaction will occur. Because activation energy is the amount of energy required for a chemical reaction to take place.
Activation energy is the minimum amount of energy needed to start a chemical reaction. 3White phosphorus has a lower activation energy than red phosphorus. The minimum amount of additional energy needed by a reacting molecule to get transformed into the product is termed activation energy.
It is not affected by a catalyst. Activation energy is best described as ___ the energy required to initiate a chemical reaction Rank the grades of coal by their relative desirabilities starting with mos desirable at the top. The activation energy of a particular reaction determines the rate at which it will proceed.
Because activation energy is the amount of energy required for a chemical reaction to take place. The maximum energy level of the reaction. The energy threshold that must be reached before a reaction can proceed and products may be formed.
The activation energy of a particular reaction determines the rate at which it. What happens as the activation energy increases. Activation energy in chemistry the minimum amount of energy that is required to activate atoms or molecules to a condition in which they can undergo chemical transformation or physical transport.
So I think its C. What is activation energy diagram. All chemical reactions including exothermic reactions need activation energy to get started.
The energy required for a reaction to proceed by breaking bonds d. When 10 mL of A is added to 10 mL of B the reaction takes twenty seconds. A type of endergonic reaction c.
The energy required for a reaction to proceed by breaking bonds. Which statement best describes the energy of activation. KJ and the reaction is.
Energy of activation The energy of activation is best described as Multiple Choice the speed at which a reaction proceeds to form products. The activation energy is the energy required to start a reaction. The activation energy is 50.
So I think its C. 4Products have higher chemical potential energy than reactants. Group of answer choices a The amount of energy lost in an exothermic reaction.
The Activation Energy can best be described as. This is the starting energy before we go on the reaction. Activation energy is the energy barrier that has to be crossed by reactants to reach transition state in order to form products.
The best statement that describes the energy of activation is that it is more quickly overcome when the temperature increases. C the energy of motion. Activation energy is needed so reactants can move together overcome forces of repulsion.
It is higher if the reaction temperature increases. Activation energy in chemistry the minimum amount of energy that is required to activate atoms or molecules to a condition in which they can undergo chemical transformation or physical transport. The difference in energy between reactants and the maximum energy.
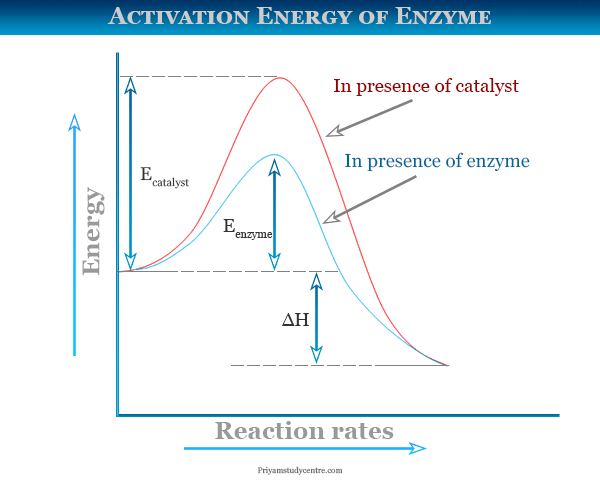
The best definition of activation energy is that activation energy is the energy required to bind a substrate to an active site option C. Enzymes are proteins that bind to a molecule or substrate to. The catalysts needed to raise a reactions rate.
As activation energy term Ea increases the rate constant k decreases and therefore the rate of reaction. Activation energy can best be described as. Which best describes the activation energy of a chemical reaction.
T he Arrhenius equation allows us to calculate activation energies if the rate constant is known or vice versa. This is the starting energy before we go on the reaction. It is more quickly overcome when the temperature increases.

Kinetics Why Is Activation Energy Drawn In A Potential Energy Diagram In Reactions Chemistry Stack Exchange
Comments
Post a Comment